
April 2026
No Pain, No Gain:
Navigating the Clinical and Commercial Realities
for Non‑Opioids
The opioid epidemic has fueled significant ongoing efforts to develop safer, non‑opioid analgesics. Journavx, a selective voltage-gated sodium channel blocker that targets Nav1.8, is the first novel non‑opioid to receive FDA approval in more than 20 years—a major breakthrough for the field. However, the journey to meaningfully reduce opioid use and provide commercially sound options for chronic pain management is only just beginning.
The FDA approval of Journavx is a breakthrough achievement, but the journey to meaningfully reduce opioid use is only just beginning.
The Dawn of a New Era with Non‑Opioids
- The opioid epidemic has fueled significant ongoing efforts to develop safer, non-opioid analgesics.
- Journavx, a selective voltage-gated sodium channel blocker that targets Nav1.8, is the first novel non-opioid to receive FDA approval in more than 20 years—a major breakthrough for the field.
- As we enter the post approval era, we explore six frontiers
that will
dictate the future of the Nav class and the non-opioid market:
- Nav1.7 vs. Nav1.8: Why did a Nav1.8 asset reach FDA approval first?
- Efficacy ceiling: Can Navs ever match opioids’ “holy grail” efficacy?
- Next generation Navs: What strategies are being adopted with the selective Nav inhibitors to improve on Journavx?
- Pain types for Navs: What are the appropriate pain subtypes for Navs? Could Navs ever make the leap from treating acute to chronic pain?
- Beyond Navs: What promising targets could follow Navs? Which non-opioid classes are nearing FDA approval?
- Reimbursement landscape: What hurdles from the Journavx launch could inform future non-opioid market access strategies?
Nav Class Overview
- Definition: Voltage-gated sodium channels (Nav) that open or close in response to stimuli, allowing sodium ions to trigger action potentials in nociceptors (pain sensing neurons).
- Subtypes: There are nine Nav isoforms (1.1-1.9) expressed in neuronal, cardiac, or skeletal tissues.
- Nav1.7-1.9: These isoforms are preferentially expressed peripherally. Inhibitors specific to these channels could provide an analgesic effect while minimizing addiction risk and cognitive impairment (sedation, dizziness) associated with centrally-acting analgesics.
Source: Xie, Yet al., (2026). International Journal of Molecular Sciences,27(3), 1180; Banh et al., (2026). CNS drugs,40(2), 165-180; Chen et al., (2025). Sodium channels as a new target for pain treatment. Frontiers in pharmacology,16, 1573254.
![]() Key Question:
Key Question:
Why did Nav1.8 assets reach FDA first?
Journavx approval highlights the technical advantages for
Nav1.8 over Nav1.7:
autonomic regulation, redundancy management, and
engine-like
action potential
Nav1.7 vs. Nav1.8
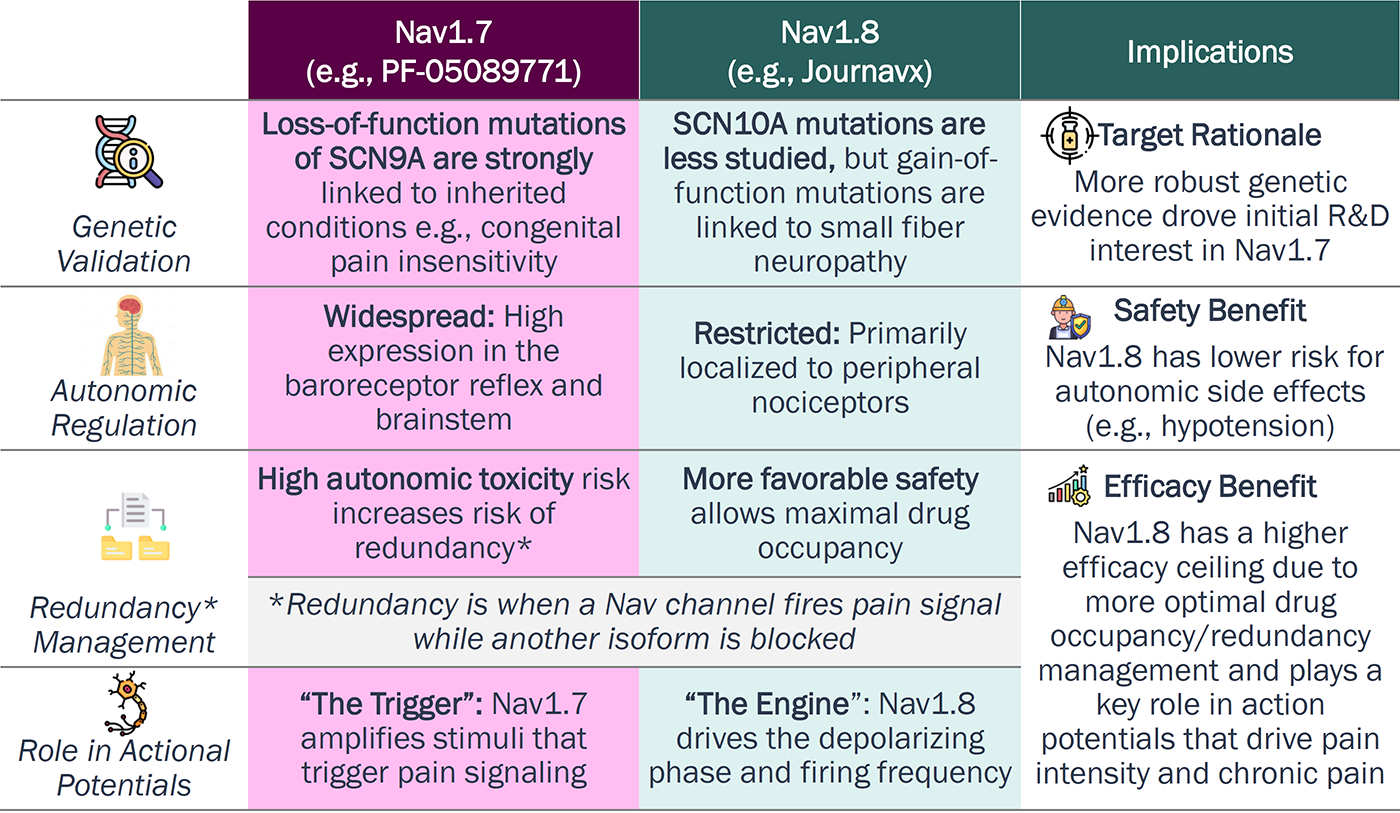
Source: Xie, Yet al., (2026). International Journal of Molecular Sciences,27(3), 1180; Banh et al., (2026). CNS drugs,40(2), 165-180; Chen et al., (2025). Sodium channels as a new target for pain treatment. Frontiers in pharmacology,16, 1573254.
![]() Key Question:
Key Question:
Can Navs ever match opioids’ “holy grail” efficacy?
Journavx has yet to consistently match or outperform opioid efficacy.
In head-to-head trials, Journavx (Nav1.8) more closely matched the efficacy of its active comparator (including opioids) than PF-05089771 (Nav1.7) in acute pain
Nav1.7 vs. Nav1.8 in Acute Post-Surgical Pain
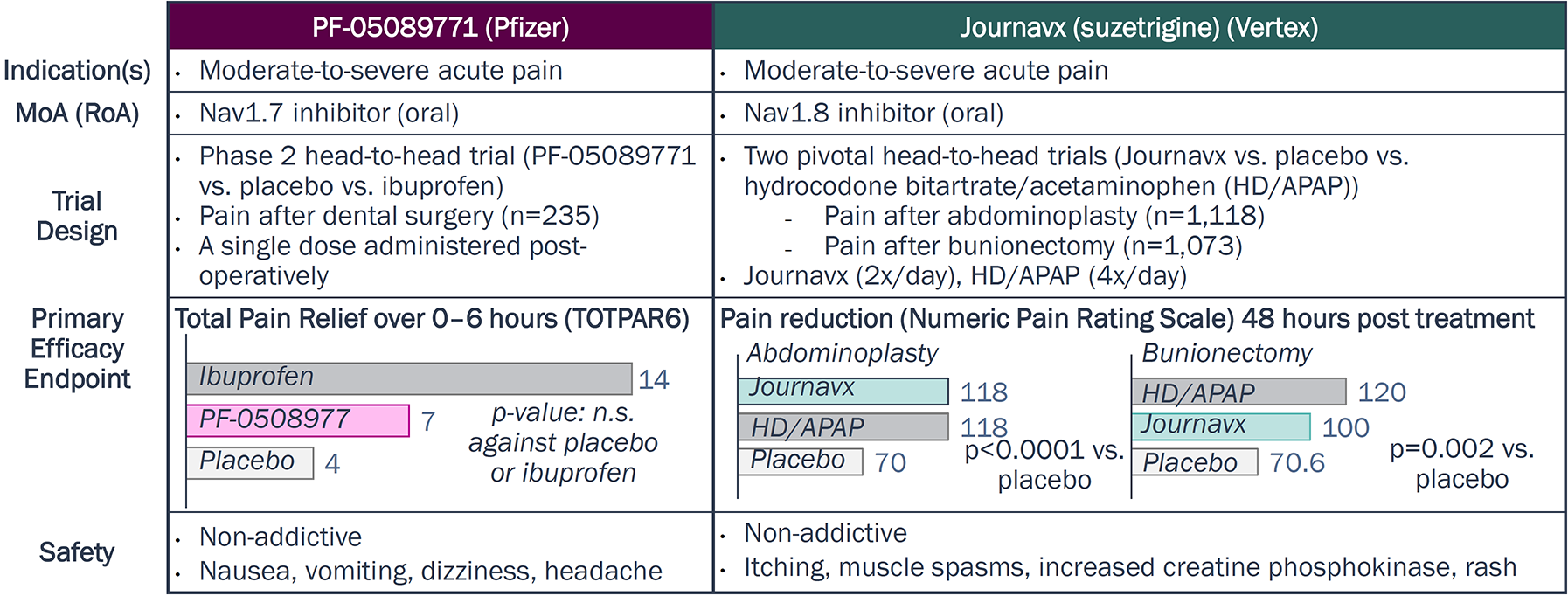
Source: Journavx package insert, Clinical trials.gov
![]() Key Questions:
Key Questions:
What are the appropriate pain subtypes for
Navs?
Could Navs ever make
the leap from treating acute to chronic pain?
Identifying the most
appropriate pain type for Navs remains a challenge,
as different pain subtypes
respond to Nav inhibitors differently.
With efficacy established for Nav1.8 in acute pain, future clinical development of Navs is largely focusing on chronic pain
Nav Inhibitors Pipeline (Marketed and Clinical Pipeline Assets for Pain in the U.S.) (n=10)
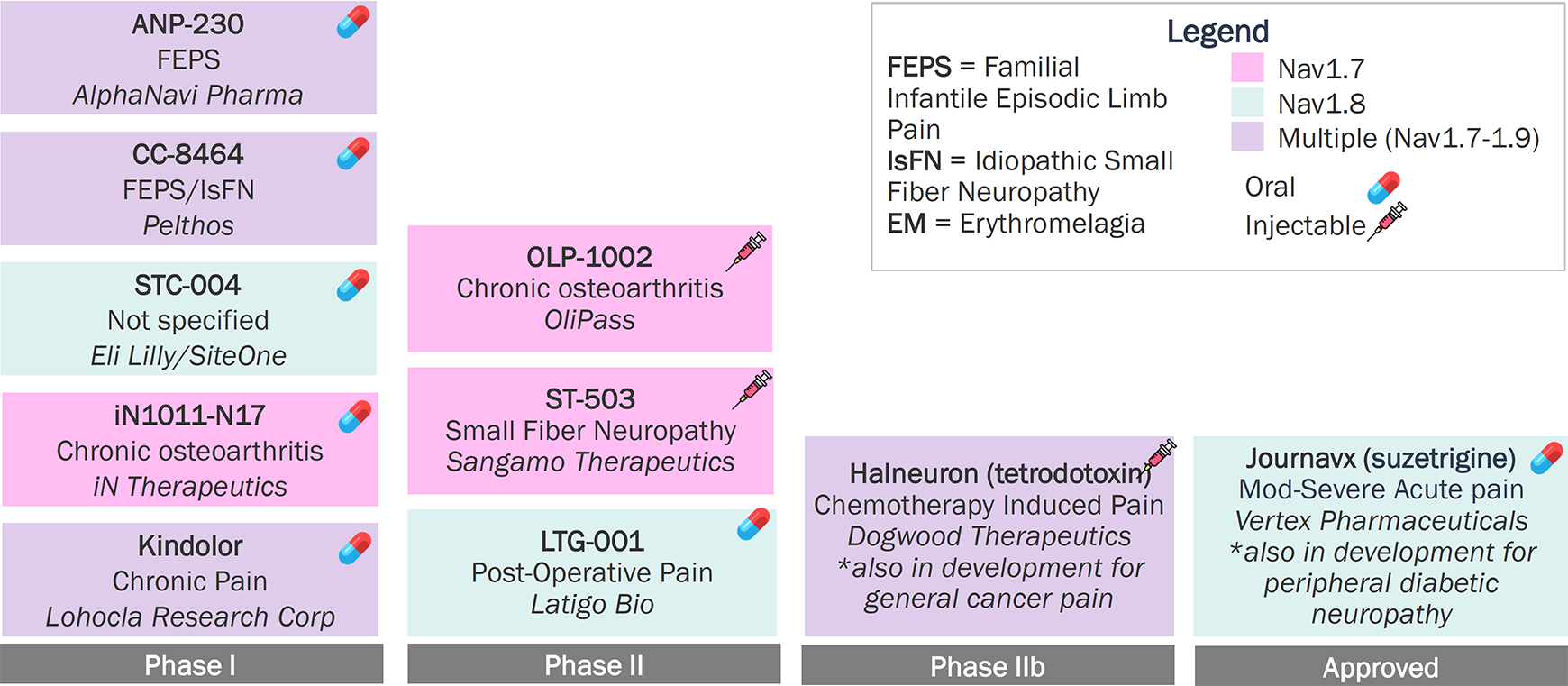
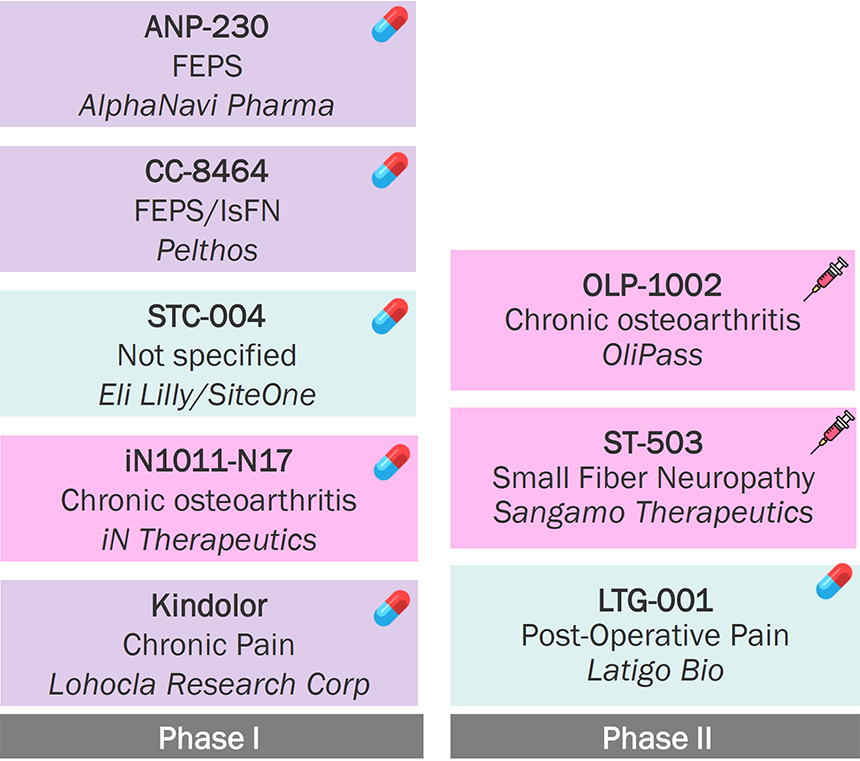
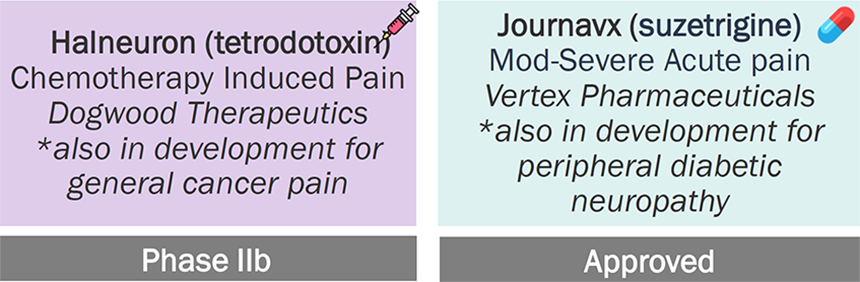

Source: Pharmaprojects; Company Websites
![]() Key Question:
Key Question:
What strategies are being adopted with the selective Nav inhibitors to improve on Journavx?
Next-generation single- and multi-subtype Nav inhibitors employ novel approaches to potentially address the limitations seen with Journavx
Strategies to Improve Navs


Source: Pharmaprojects; Company Websites
![]() Key Questions:
Key Questions:
What promising targets could follow Navs?
Which non-opioid classes
are
nearing FDA approval?
Assets with readouts in 2026 are either targeting chronic pain indications or exploring novel mechanisms.
The non-opioid landscape extends well beyond Nav channels to include diverse mechanisms such as GPCRs, endocannabinoids, and neuroimmune modulators
Non-Opioids Pipeline in the U.S. (n=52)
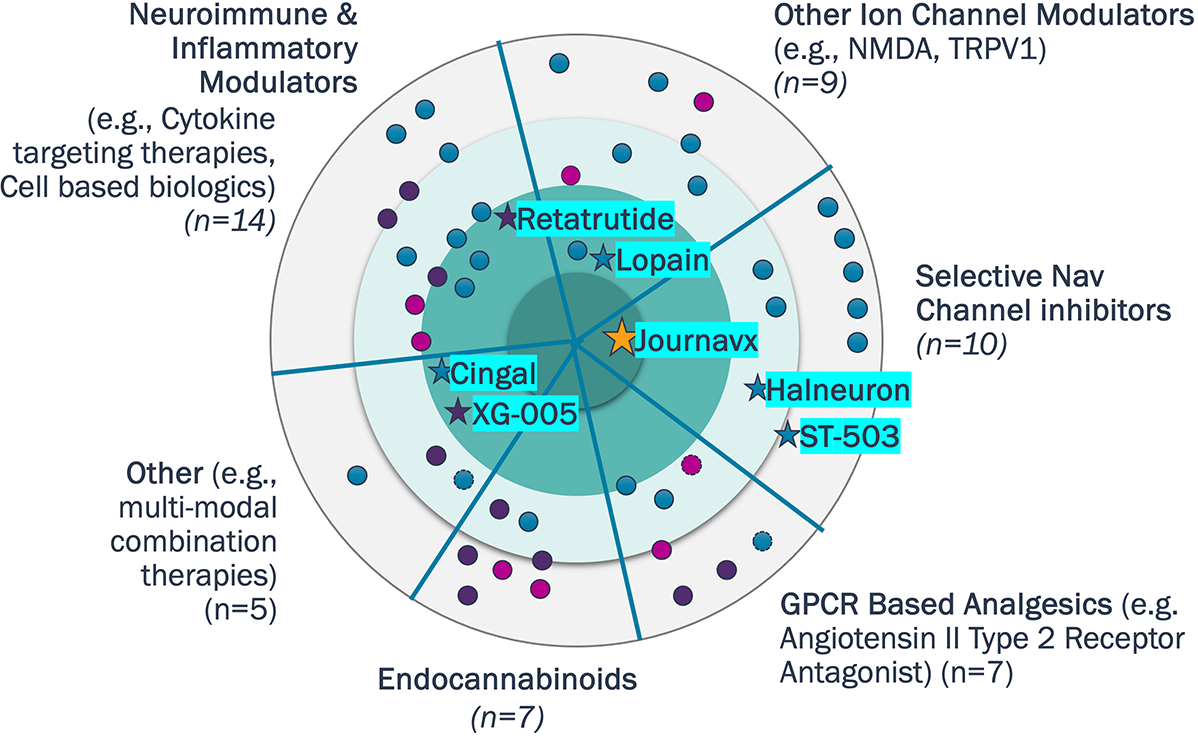

Source: Pharmaprojects; Company Websites
![]() Key Question:
Key Question:
What commercial and reimbursement hurdles from the Journavx launch could inform future market access strategies for other non-opioids?
Journavx’s premium pricing, tiered access, and evidence generation strategy may provide a roadmap for reimbursement and market entry for future non-opioids
Commercial Outlook for Journavx and Other Non-Opioids
Journavx U.S. Eight-months Launch Performance
- $60M Revenue ($27M in Q4 2025 alone; revenue missed forecast expectations of $33M).
- 420K patients treated (550K prescriptions written by 35K HCPs).
- ~200M lives covered (coverage in 21 states to date; Journavx is on 100+ healthcare systems and 950 hospital formularies).
- Cost: ~$31/day (NSAIDs/opioids cost <$2 /day).
Key Challenges
Select Strategies
Premium Price Justification
Journavx’s lack of superior efficacy over NSAIDs/opioids in
head-to-head trials makes its price unjustifiable especially as a first
line therapy.
Real World Evidence
Vertex’s conducted Phase IV
to ease payer restrictions. Journavx led to a 90.9% opioid-free recovery
(vs. the 10% with NSAIDs only).
High Tier Placement/
Non-Preferred Brand
Status
Higher costs and non-superiority data relegate
Journavx to non-preferred status, forcing mandatory step edits through
generic NSAIDs and/or opioids.
Health Economics and Outcomes Research
Vertex cites
ICER’s $30K cost burden associated with each opioid use disorder
(OUD) case; ~85K patients will develop an OUD annually.
Source: Vertex Q4 Earnings Press Release and Transcript (Feb 2026); Manalac, T (2025, February 24) BioSpace; Pretorius, R. (2025, March 17) Syenza News; Bell, J. (2025, November 4), BioPharma Dive; Bell, J. (2025, March 6) BioPharma Dive
Abbreviations: NSAIDs = Non-steroidal anti-inflammatory drugs; ICER = Icer Cost Effectiveness Analysis.